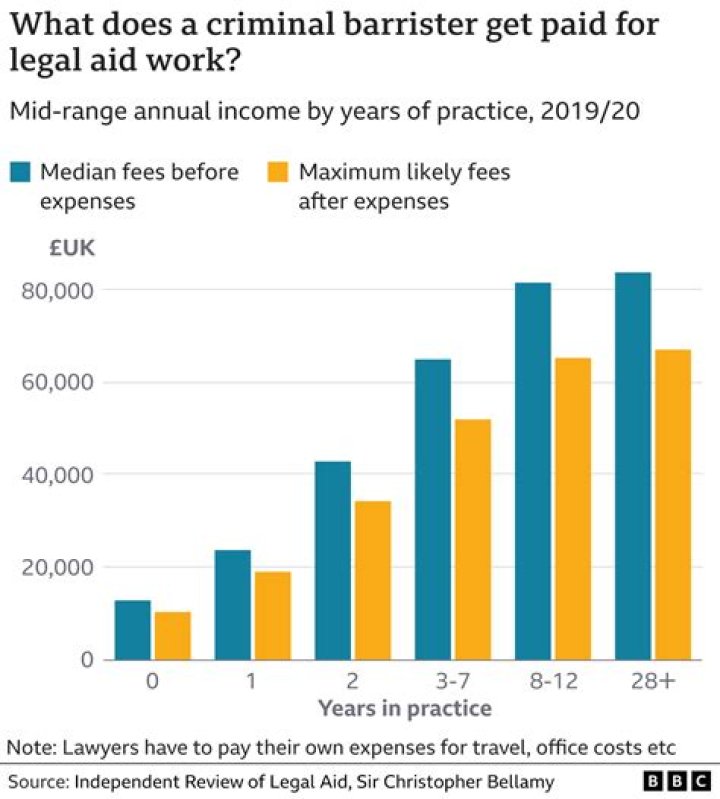
Iodine(III) Bromide. Alias: Iodine Tribromide. Formula: IBr3..
Also know, what is the chemical name of IBr3?
Iodine tribromide
| PubChem CID: | 5187573 |
| Molecular Formula: | IBr3 or Br3I |
| Chemical Names: | Iodine tribromide 7789-58-4 tribromo- EINECS 232-176-1 AC1NPZ8Q More |
| Molecular Weight: | 366.62 g/mol |
| Dates: | Modify: 2019-08-24 Create: 2005-09-26 |
Secondly, what is the hybridization of IBr3? Hybridization of IBr3 is sp3d. It's geometry is trigonal bipyramidal and shape is trigonal planar (due to presence of 2 lone pairs).
Regarding this, what is the molecular geometry of IBr3?
The molecular geometry of Br3 is T-shaped with an asymmetric charge distribution on the central atom. Therefore this molecule is polar. This is somewhat of a rare molecule (perhaps difficult to manufacture).
Which is the formula for iodine Tribromide?
Iodine tribromide Formula - Br3I - Over 100 million chemical compounds | Mol-Instincts.
Related Question Answers
How many lone pairs does IBr3 have?
2 lone pairs
Is BrCl3 polar or nonpolar?
BrCl3: the molecule has a T-shaped molecular geometry which could make it polar, but the Br-Cl electronegativity difference is 0.2 which makes it a covalent bond. CS2: the molecule has a linear geometry and the electronegativity difference between C and S is 0, so this one is not polar either.Why is ibr3 polar?
Decision: The molecular geometry of Br3 is T-shaped with an asymmetric charge distribution on the central atom. Therefore this molecule is polar. This is somewhat of a rare molecule (perhaps difficult to manufacture).Is XeF4 polar or nonpolar?
XeF4 Polarity – Is XeF4 Polar or Nonpolar? As we can see that the XeF4 molecular geometry has the symmetric distribution of electrons and they make a formation in the single square plane. So, we can say that the XeF4 is nonpolar.What is the hybridization of bromine in BrF5?
Hybridization of BrF5 (Bromine Pentafluoride) It is a colourless liquid and has properties like strong odour, high toxicity, and is corrosive in nature. This liquid reacts explosively when it comes in contact with organic materials. The hybridization that takes place in BrF5 is sp3d2.Is pf2cl3 polar or nonpolar?
The molecular geometry of PF2Cl3 is trigonal bipyramidal with symmetric charge distribution around the central atom. Therefore this molecule is nonpolar. This is somewhat of a rare molecule, but here is one like it: Phosphorus Pentachloride on Wikipedia.Is XeF2 polar or nonpolar?
In XeF2 molecule, two fluorine atoms are arranged symmetrically on the outside with the central atom Xenon in the middle. There is no net dipole moment in the compound due to the arrangement of the valence electrons in symmetry. Hence Xenon Difluoride is nonpolar as there is no polarity observed in the molecule.What shape is ClF3?
ClF3 molecular geometry is said to be a T-shaped. It acquires such shape because of the presence of two lone pairs which take up equatorial positions and there are greater repulsions. The electron geometry of chlorine trifluoride is trigonal bipyramidal with a 175° F-Cl-F bond angle.What is the bond angle of T shaped?
120
o.
How do you determine hybridization?
You can find the hybridization of an atom by finding its steric number: The steric number = the number of atoms bonded to the atom + the number of lone pairs the atom has. If the steric number is 4, the atom is sp3 hybridized. If the steric number is 3, the atom is sp2 hybridized.Is ClF3 planar?
Chlorine trifluoride(ClF3) has 5 regions of electron density around the central chlorine atom (3 bonds and 2 lone pairs). Due to the T-shaped structure between the central molecule and the three bond pair, the molecule is planar.Why is ClF3 not trigonal planar?
ClF3 Chlorine Trifluoride These are arranged in a trigonal bipyramidal shape with a 175° F(axial)-Cl-F(axial) bond angle. The two lone pairs take equatorial positions because they demand more space than the bonds. The result is a T-shaped molecule.What is the electron pair geometry for P in pf5?
The molecular geometry of PF5 is trigonal bipyramidal with symmetric charge distribution.What is the polarity of ClO3?
3 pts) ClO3. – is polar because the Cl – O bonds are polar and they are arranged so that the Cl ends are at the top of a. pyramid and the O ands are at the bottom of a pyramid.What is the polarity of ClF3?
The molecular geometry of ClF3 is T-shaped with asymmetric charge distribution around the central atom. Therefore ClF3 is polar.What is the electronic geometry of bf3?
The geometry of the BF3 molecule is called trigonal planar. The fluorine atoms are positioned at the vertices of an equilateral triangle. The F−B−F angle is 120o, and all four atoms lie in the same plane.What is the bond angle of ClF3?
ClF3 shows trigonal planar geometry because of lone pair on Cl atom. Lone pair -bond pair repulsion causes the bond angle to shrink and so bond angle will be 107.5° instead of 109.5°( for perfect tetrahedral geometry).What type of hybridization occur in BrF3?
Bromine belongs to group 17 , with 7 electron in the outermost shell. BrF3 has a Bent T shaped structure with Br as a central atom bonded with three F atoms (three bond pairs) and two lone pairs. Hence , the hybridization would be sp3d.Is T shaped polar?
T shaped molecules are always polar. In both cases (octahedral and trigonal bipyramidal), the bond angles will be slightly less than 90 and 180 due to lone pair-bond pair repulsion.